Conclusion AXn is going to be nonpolar. In VSEPR theory the lone pair forces the molecular geometry of SF4 into a see-saw shape.

Science Coverage Is Asf5 Polar Or Nonpolar In 2021 Molecular Geometry Molar Mass Octet Rule
Nonpolar but contains at least one polar covalent bond D.

Xecl4 polar or nonpolar. But the other two S-F dipoles are pointing down. It is a molecule with octahderal electron pair geometry and square planar molecular geometry. Ok as far as molecular geometry goes you can easily tell if the molecule is going to be polar or non polar.
Asked by Wiki User. The molecule is nonpolar because the two Kr-F bond dipoles cancel each other. They cancel each other.
The two non bonded pairs of electrons are set at the perpendicular axis. Electrons available 1Xe 5F - 1 8 35 - 1 42 Five Xe-F bonds account for 40 valence electrons but you actually have 42 electrons available. What is the value of the smallest bond angle in xecl4.
What are the approximate bond angles in XeCl4. Ill tell you the polar or nonpolar list below. Boron Trihydride - YouTube.
AX3E trigonal pyramidal polar AX2E2 bent polar. In chemistry polarity is defined as a physical property of matter relating to the unequal dispersion of partial charges between intermolecular atoms. Is BH3 Polar or Non-polar.
List molecules polar and non polar. See-saw molecular geometry E. Answer SCl4 Sulfur tetrachloride is Polar.
The S-O bond dipoles do not cancel so SO 2 is polar. Be-H dipoles are equal and point in opposite directions. Polar but contains no polar bonds B.
What is polar and non-polar. Select all that apply. AX4E seesaw polar AX3E2 t shaped polar AX2E2.
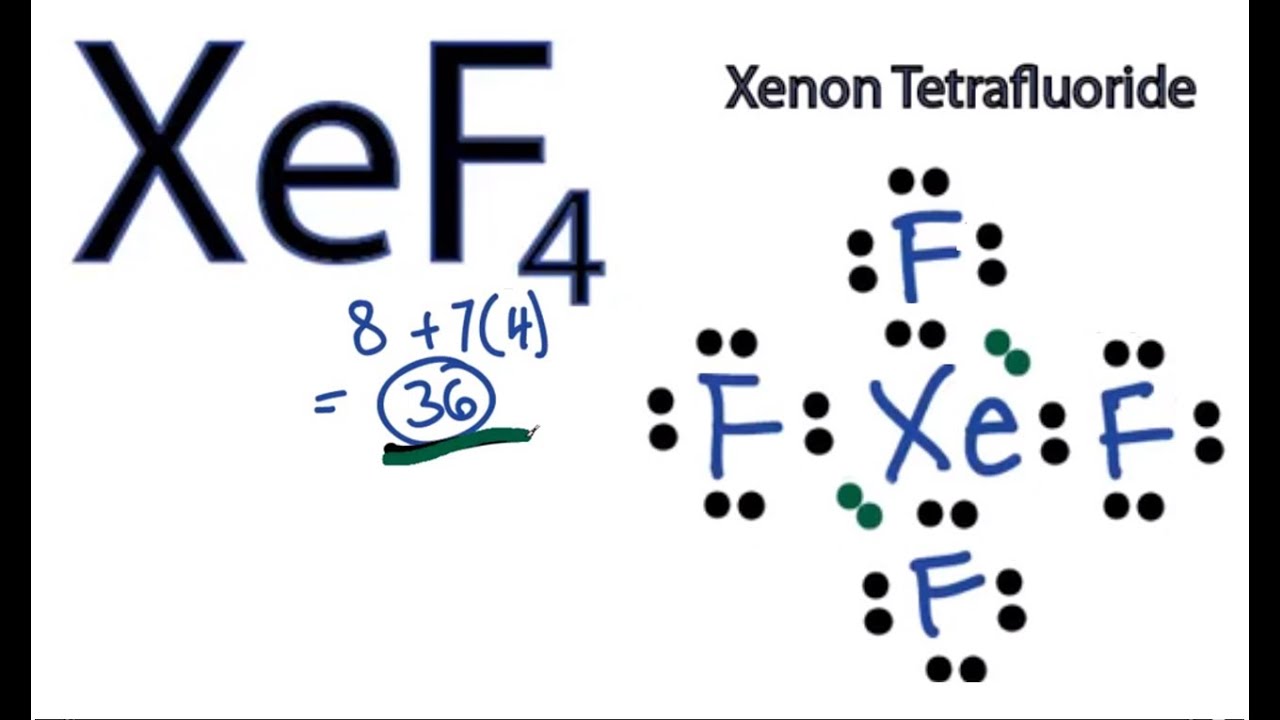
Learn to determine if XeF4 is polar or nonpolar based on the Lewis Structure and the molecular geometry shapeWe start with the Lewis Structure and then us. Polar or Nonpolar. The molecular structure of xenon tetrafluoride is square planar with six uniformly placed sp 3 d 2 orbitals forming 90-degree angles.
It is polar because the geometry does not cancel out the charge of this molecule and the electronegativity difference is greater than 5. The molecule is therefore non-polar. - A SF4 - A XeCl4 - A CH2F2 - BC13 - A sp - sp2 - A sp3 A.
The 4 F atoms are arranged around the Xe atom with bond angles of 90 degree. QUESTION 11 Match the following. A 90 degrees b 1095 degrees c 120 degrees d 180 degrees Identify the molecular geometry of XeCl4 a tetrahedral b trigonal bipyramidal c T-shaped d linear e see-saw f octahedral g square pyramidal h bent i trigonal pyramidal.
The individual Xe-F bonds are polar due to unequal electronegativity of Xe and F atoms but the net vector sum of the polarities of Xe-F bonds is zero as they cancel out each other. Some molecular shapes are seen as perfect and will always lead to a non-polar molecule overall. Is XeF4 Polar or Nonpolar.
If you want to quickly find the word you want to search use Ctrl F then type the word you want to search. Although the Xe-F bonds themselves are polar covalent the symmetrical arrangement of these bonds results no net dipole moment cancellation of the polar vectors. Although Carbon and hydrogen have an electronegativity difference of 035 the dipole gets canceled out due to the symmetry making the C6H12.
XeF4 has a symmetrical square planar shape. Priya G N Meritnation Expert added an answer on 25313 XeF4 is non-polar based on its symmetric square planar structure and also XeF4 has 2 lone pairs on XeThe lone pairs are opposite to each other and their attractionrepulsion is cancelled. BeH 2 is nonpolar.
The molecule XeF4 known as xenon tetrafluoride is nonpolar. Transcribed Image Textfrom this Question. XeCl4 molecule is a polar.
In chemistry polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole or multipole moment. XeCl4 molecule is a polar. Octahedral electron geometry.
Answer to Determine whether each molecule is polar or nonpolar. Xenon tetrafluoride XeF4 is a non-polar chemical compound owing to its symmetrical square planar structure. How come SnCl2 is polar.
Answer XeCl4 is Nonpolar What is polar and non-polar.

Is Xef4 Polar Or Non Polar Xenon Tetrafluoride Youtube

Science Coverage Is No2 Polar Or Nonpolar Electron Affinity Covalent Bonding Molecular Geometry

Xef4 Lewis Structure How To Draw The Lewis Structure For Xef4 In 2021 Octet Rule Lewis Noble Gas

Science Coverage Is Clo3 Polar Or Nonpolar In 2021 Molecular Geometry Covalent Bonding Oxidation State

Science Coverage Is Hcl Polar Or Nonpolar Covalent Bonding Molecular Geometry Solubility

Is Xef4 Polar Or Nonpolar Xenon Tetrafluoride Youtube
Is Xef4 Polar Or Non Polar Xenon Tetrafluoride Youtube

Is Xecl4 Polar Or Nonpolar Xenon Tetrachloride Youtube

Is Xef4 Polar Or Non Polar Xenon Tetrafluoride Youtube

Is Scn Polar Or Nonpolar Covalent Bonding Molecular Geometry Solubility

Xef4 Molecular Geometry Xenon Tetrafluoride Is A Chemical Compound With Chemical Formula Xef4 It Is Produced By Molecular Geometry Molecular Chemical Formula

Science Coverage Is Hi Polar Or Nonpolar Electron Affinity Covalent Bonding Redox Reactions
Is Xef4 Polar Or Nonpolar Quora

Is Xef4 Polar Or Non Polar Xenon Tetrafluoride Youtube

Is Xecl4 Polar Or Nonpolar Xenon Tetrachloride Youtube

Science Coverage Is Hf Polar Or Nonpolar Electron Affinity Covalent Bonding Molecular Geometry


Post a Comment